Side effects during subcutaneous immunotherapy (SCIT) in children: severe systemic side effects in 0.03% of all treatments
Allergen-specific immunotherapy (AIT) is the only causal form of therapy for IgE-mediated allergic diseases:
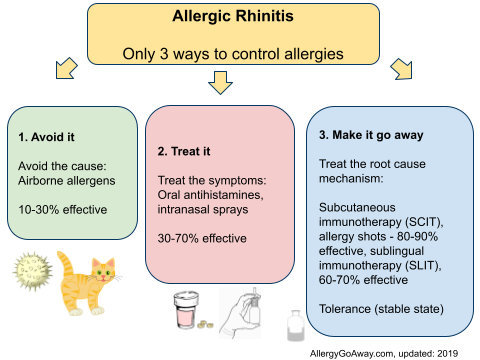
Allergic Rhinitis - Only 3 ways to control allergies (click to enlarge the image).
Subcutaneous immunotherapy (SCIT) is considered safe and well tolerated in adults, yet there is less evidence of safety in the pediatric population.
This prospective study included 581 pediatric patients in 18 study centers in Germany. The study included 8,000 treatments and 10,000 injections.
Local reactions
55% of the patients experienced immediate local side effects at least once. Delayed local side effects were seen in 56%.
Systemic reactions
Immediate systemic adverse reactions occurred in 2% of patients. However, 7% experienced delayed systemic side effects.
Severe systemic side effects (grade III in the classification of Ring and Messmer) were seen in only 0.03% of all treatments, all appearing within 30 minutes after the injections.
References:
Tophof, M. A., Hermanns, A., Adelt, T., Eberle, P., Gronke, C., Friedrichs, F., Knecht, R., Mönter, E., Schöpfer, H., Schwerk, N., Steinbach, J., Umpfenbach, H.-U., Weißhaar, C., Wilmsmeyer, B. and Bufe, A. (), Side effects during subcutaneous immunotherapy (SCIT) in children with allergic diseases. Pediatr Allergy Immunol. Accepted Author Manuscript. doi:10.1111/pai.12847
http://onlinelibrary.wiley.com/doi/10.1111/pai.12847/abstract
Allergic Rhinitis - Only 3 ways to control allergies (click to enlarge the image).
Subcutaneous immunotherapy (SCIT) is considered safe and well tolerated in adults, yet there is less evidence of safety in the pediatric population.
This prospective study included 581 pediatric patients in 18 study centers in Germany. The study included 8,000 treatments and 10,000 injections.
Local reactions
55% of the patients experienced immediate local side effects at least once. Delayed local side effects were seen in 56%.
Systemic reactions
Immediate systemic adverse reactions occurred in 2% of patients. However, 7% experienced delayed systemic side effects.
Severe systemic side effects (grade III in the classification of Ring and Messmer) were seen in only 0.03% of all treatments, all appearing within 30 minutes after the injections.
References:
Tophof, M. A., Hermanns, A., Adelt, T., Eberle, P., Gronke, C., Friedrichs, F., Knecht, R., Mönter, E., Schöpfer, H., Schwerk, N., Steinbach, J., Umpfenbach, H.-U., Weißhaar, C., Wilmsmeyer, B. and Bufe, A. (), Side effects during subcutaneous immunotherapy (SCIT) in children with allergic diseases. Pediatr Allergy Immunol. Accepted Author Manuscript. doi:10.1111/pai.12847
http://onlinelibrary.wiley.com/doi/10.1111/pai.12847/abstract